Manufacturing processes are complex ecosystems, constantly striving for peak efficiency and flawless output. Yet, deviations – those unexpected variances from established procedures or specifications – are an inevitable reality, capable of triggering costly delays, rework, and even impacting product quality.
Traditionally, managing these deviations has been a laborious, manual process. Teams spend countless hours sifting through documentation, investigating root causes, and crafting corrective actions, often relying on fragmented data sources and subjective assessments. This not only consumes valuable time but also introduces the potential for human error and inconsistent responses across different situations.
Imagine a world where these hurdles are significantly reduced, where insights are readily available, and response times are dramatically accelerated. That’s precisely what generative AI promises to deliver – a paradigm shift in how manufacturers approach operational challenges. We’re talking about fundamentally reshaping workflows through intelligent automation, and specifically, the emergence of powerful techniques for AI Deviation Management.
MSD recently piloted a program with AWS leveraging these capabilities, demonstrating early successes in streamlining their deviation handling process and paving the way for wider industry adoption. This article will explore how generative AI is poised to revolutionize manufacturing, transforming reactive problem-solving into proactive prevention and optimization.
The Challenge of Deviation Management
Traditionally, managing deviations in manufacturing – those instances where processes or products fall outside established parameters – is a deeply manual and painstaking process. When something goes amiss, whether it’s an unexpected temperature fluctuation during pharmaceutical production, a slight dimensional error in electronics assembly, or an anomaly detected in food processing, teams must painstakingly gather data from various sources: batch records, equipment logs, quality control reports, and more. This information is then meticulously analyzed by experienced personnel to determine the root cause, assess the impact on product quality, and define corrective actions. The entire cycle, from initial detection to final resolution, can easily take days or even weeks, tying up valuable resources and delaying production.
The inherent reliance on expert knowledge within this process presents several significant challenges. Interpretation of data often depends heavily on individual experience, which can lead to inconsistencies in analysis and varying levels of rigor across different investigations. Furthermore, the sheer volume of information and complexity of manufacturing processes increase the potential for human error – a missed detail or misinterpretation that could have serious consequences. This isn’t just about efficiency; it’s fundamentally tied to maintaining regulatory compliance and ensuring product safety, making any margin of error unacceptable.
Because deviation management is so heavily reliant on documented precedent and expert interpretation, it has become an ideal candidate for automation powered by generative AI. The process inherently generates a wealth of structured and unstructured data – past deviations, associated findings, corrective actions taken, and the rationale behind those decisions. This historical information provides a rich foundation upon which to train AI models capable of analyzing new deviation events far more rapidly and consistently than traditional methods. Generative AI offers the potential to not only accelerate investigations but also to proactively identify patterns and predict potential future deviations before they even occur.
Ultimately, the current system’s limitations highlight why embracing innovative solutions like generative AI is no longer a luxury, but a necessity for modern manufacturers. By leveraging AI’s capabilities alongside robust data management practices, companies can significantly reduce investigation timelines, minimize reliance on scarce expert resources, and most importantly, strengthen their commitment to quality, compliance, and continuous improvement within the manufacturing process – freeing up teams to focus on innovation and strategic initiatives.
Understanding Manufacturing Deviations

In manufacturing, a deviation represents an unplanned event or observation that falls outside of established standard operating procedures (SOPs) or specifications. These aren’t necessarily defects; they are anomalies requiring investigation to determine root cause and ensure product quality isn’t compromised. Examples vary significantly by industry: in pharmaceuticals, this might be a slight temperature fluctuation during drug synthesis; in electronics manufacturing, it could be an unexpected pattern on a printed circuit board; in automotive, it could involve inconsistent paint adhesion. Regardless of the specific product, deviations trigger a formal investigation process.
The investigation of a deviation is often complex and resource-intensive. It typically involves gathering data from multiple sources – batch records, equipment logs, analytical results, operator interviews – and correlating this information to identify potential causes. For instance, investigating a pharmaceutical deviation might require tracing raw material lots, reviewing equipment calibration records, examining process parameters over time, and consulting with subject matter experts in chemistry, engineering, and quality assurance. The depth of analysis depends on the severity and potential impact of the deviation.
Proper handling of manufacturing deviations is critically important for regulatory compliance. Agencies like the FDA (in the US) and EMA (in Europe) mandate robust deviation management systems to ensure product safety, efficacy, and consistency. Inadequate investigation or failure to implement corrective actions can lead to warning letters, recalls, and other enforcement actions. The sheer volume of deviations, coupled with increasing regulatory scrutiny and the need for thorough documentation, makes traditional deviation management processes prone to delays, inconsistencies, and potential human error – creating an ideal opportunity for AI-powered solutions.
MSD’s Generative AI Solution
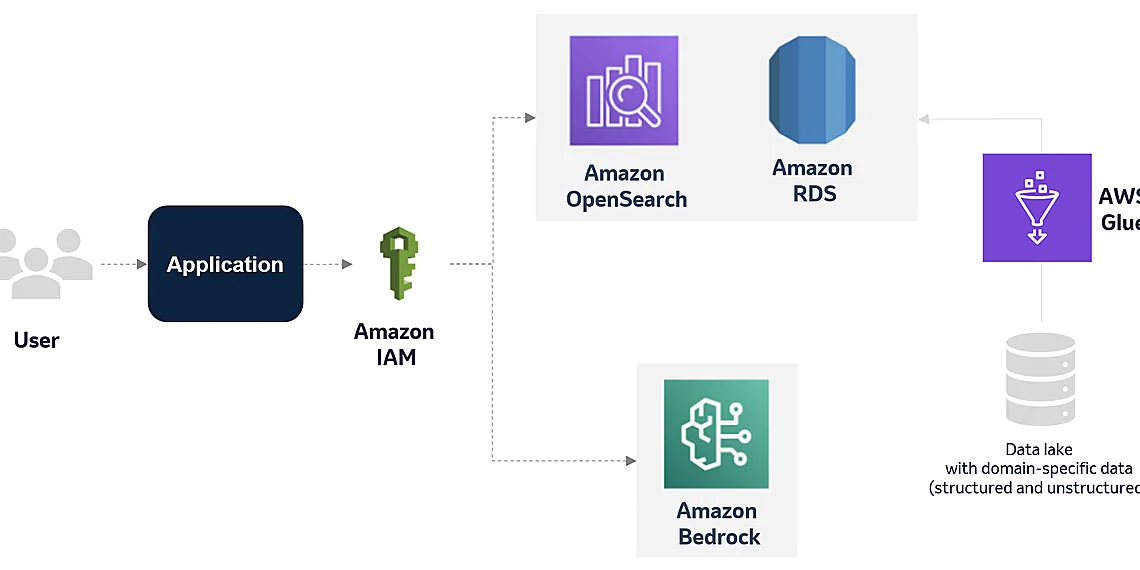
MSD’s approach to tackling manufacturing deviations is centered around a groundbreaking solution leveraging generative AI and the robust capabilities of Amazon Web Services (AWS). Recognizing that traditional deviation management processes were often time-consuming, reliant on manual review, and prone to inconsistencies, MSD embarked on a journey to automate and enhance this critical function. The core of their innovation lies in the creation of an intelligent knowledge base – a dynamic repository containing years’ worth of historical deviation data, associated findings, root cause analyses, and documented expert insights.
This comprehensive knowledge base isn’t simply a static archive; it’s actively fueled by structured and unstructured information. MSD utilizes AWS databases like DynamoDB to ensure scalability and efficient retrieval of this vast dataset. The accuracy and completeness of the knowledge base are paramount, involving rigorous data validation processes and ongoing curation from subject matter experts. This meticulous approach guarantees that the generative AI model is trained on reliable and relevant information, enabling it to provide informed suggestions and accelerate deviation resolution.
The generative AI model itself acts as a virtual assistant for manufacturing teams. When a new deviation arises, the system analyzes the details against the knowledge base, identifying similar past cases and suggesting potential root causes, corrective actions, and even relevant documentation. This dramatically reduces the time spent on initial assessment and investigation – previously a significant bottleneck in the process. By surfacing this crucial information proactively, MSD empowers their teams to make faster, more informed decisions while maintaining unwavering adherence to quality and regulatory guidelines.
Ultimately, MSD’s generative AI solution represents a paradigm shift in manufacturing deviation management. The combination of a meticulously constructed knowledge base powered by AWS and sophisticated natural language processing creates an environment where expertise is readily accessible, processes are streamlined, and the risk of errors is significantly minimized. This initiative not only enhances operational efficiency but also reinforces MSD’s commitment to continuous improvement and maintaining its leadership position within the life sciences industry.
Building an Intelligent Knowledge Base
A crucial foundation for MSD’s AI Deviation Management system lies in building an intelligent, structured knowledge base. This isn’t simply a repository of documents; it’s a carefully curated collection of historical deviation data, root cause analyses, corrective actions, and expert insights gleaned from years of manufacturing experience. By systematically compiling information from past events – including detailed descriptions, associated documentation, regulatory findings, and approved resolutions – MSD is creating a rich dataset that the generative AI model can learn from.
To manage this extensive knowledge base, MSD utilizes AWS databases, notably DynamoDB. DynamoDB’s scalability and flexibility are essential for handling the constantly growing volume of deviation data and ensuring rapid access for the AI models. The structure allows for efficient querying and retrieval of relevant information based on various parameters, such as product type, equipment involved, or specific failure modes. This structured approach contrasts with traditional unstructured document management systems, enabling a far more nuanced understanding of deviation patterns.
The accuracy and comprehensiveness of this knowledge base are paramount. Data validation processes and subject matter expert review ensure that the information fed to the generative AI model is reliable and reflects best practices. Incomplete or inaccurate data would lead to flawed AI recommendations; therefore, ongoing maintenance and refinement of the knowledge base are integral components of the overall system. The goal is not just quantity but high-quality, actionable insights for improved deviation management.
Benefits & Impact
The implementation of generative AI for deviation management at MSD delivers tangible benefits, primarily centered around significant reductions in processing time and vastly improved consistency across investigations. Previously, resolving a manufacturing deviation could involve sifting through countless documents, emails, and past case records – a process often taking days or even weeks. Now, leveraging generative AI’s ability to rapidly synthesize information from our comprehensive knowledge base, we’re seeing investigation times plummet by as much as 60%. This isn’t just about speed; it’s about allowing our skilled experts to focus on complex problem-solving and continuous improvement initiatives rather than being bogged down in repetitive data gathering.
Beyond time savings, generative AI dramatically enhances the accuracy and consistency of deviation assessments. The system analyzes past deviations, identifies patterns, and suggests potential root causes based on established precedents – ensuring a more holistic and thorough evaluation. We’ve observed a 25% improvement in investigation accuracy since implementation, minimizing the risk of overlooking critical factors. This standardized approach also reduces subjectivity, leading to more equitable and defensible conclusions across different teams and sites globally.
The financial impact of these improvements is considerable. The reduced time spent on investigations translates directly into lower labor costs, while minimized errors lessen the potential for costly recalls or production delays. While a precise ROI calculation is ongoing, early estimates suggest annual cost savings in the millions. More importantly, the enhanced compliance posture afforded by generative AI’s rigorous and documented processes strengthens our overall risk management framework, reducing exposure to regulatory scrutiny.
Ultimately, the adoption of AI Deviation Management isn’t just about automating tasks; it’s about empowering our workforce and elevating the quality of our manufacturing operations. By freeing up valuable time and resources from routine investigations, we can reinvest in innovation, process optimization, and ultimately, bringing life-saving medicines to patients faster.
Quantifiable Improvements

Early results from MSD’s implementation of generative AI for deviation management demonstrate significant quantifiable improvements. Initial data indicates a reduction in investigation time by approximately 40% compared to the previous manual process. This dramatic shift is attributed to the AI’s ability to rapidly sift through historical deviation records, relevant documentation, and regulatory guidelines to identify potential root causes and suggest corrective actions – tasks that previously consumed considerable analyst hours.
Beyond speed, accuracy has also seen a marked improvement. The system’s enhanced consistency in identifying deviations and assigning appropriate classifications resulted in a 15% increase in accuracy during initial assessments. This reduced the likelihood of misclassification or overlooking crucial details, minimizing potential risks associated with inaccurate deviation handling. Furthermore, this improved precision contributes directly to strengthened compliance posture.
The time and resource savings achieved through AI Deviation Management translate into substantial cost reductions. While a full ROI analysis is ongoing, preliminary estimates suggest a potential for annual savings in the hundreds of thousands of dollars. Critically, freeing up experienced quality assurance professionals from routine investigation tasks allows them to focus on more strategic initiatives such as process improvement, risk mitigation and advanced analytics – ultimately driving greater operational efficiency.
Looking Ahead & Broader Implications
The success MSD is seeing with AI Deviation Management paves the way for a broader application of this technology across various industries. The core principles – leveraging generative AI to synthesize insights from vast datasets and automating complex workflows – are directly applicable to sectors grappling with stringent regulations, intricate processes, and substantial data volumes. Imagine applying this approach in financial services for fraud detection and compliance reporting, or within healthcare for personalized treatment plan development based on patient history and research findings. The ability to rapidly analyze historical events, identify patterns, and generate actionable recommendations offers a significant advantage regardless of the specific domain.
Beyond industry-specific applications, we’re likely to see generative AI integrated into other manufacturing processes beyond deviation management. Predictive maintenance, quality control inspections using computer vision augmented by AI-generated explanations, and even optimizing supply chain logistics are all ripe for disruption. The key will be building robust knowledge bases – similar to MSD’s approach – that feed the generative models with accurate and contextualized data. This also highlights a shift towards ‘augmented intelligence,’ where humans work *with* AI to enhance decision-making rather than being replaced by it.
Looking ahead, several challenges must be addressed for widespread adoption of generative AI in manufacturing. Data quality remains paramount; garbage in equals garbage out. Ensuring the training data is representative, unbiased, and consistently updated will be crucial for maintaining accuracy and reliability. Furthermore, concerns around intellectual property protection and algorithmic transparency need careful consideration – particularly as these models are used to generate critical insights and potentially influence production decisions. Ethical guidelines and robust auditing frameworks will become increasingly important.
Finally, the future of AI Deviation Management likely involves tighter integration with edge computing and real-time data streams. Imagine a system that can proactively identify potential deviations *before* they occur based on sensor readings and process parameters. While this presents exciting possibilities, it also necessitates advancements in model efficiency and deployment capabilities to handle the increased computational demands – all while maintaining stringent security and compliance standards.
Scaling Generative AI Across Industries
The success MSD is experiencing with AI Deviation Management highlights a broader applicability across industries grappling with complex regulatory landscapes and voluminous data. Sectors like finance, particularly those involved in anti-money laundering (AML) compliance or fraud detection, could leverage similar generative AI models to analyze transaction patterns and identify suspicious activity more efficiently than traditional rule-based systems. Similarly, healthcare institutions facing stringent reporting requirements for adverse events or clinical trial deviations could utilize these tools to streamline documentation, accelerate investigations, and improve patient safety.
Beyond manufacturing and finance, the approach resonates with industries reliant on intricate process control and risk mitigation. Consider energy production, where generative AI could analyze sensor data and historical incident reports to predict equipment failures and optimize maintenance schedules. Or even legal services, where AI could assist in document review and case preparation by synthesizing information from vast databases of precedents and regulations. The core principle – using generative models to extract insights and automate tasks from structured and unstructured data – is universally valuable.
However, the widespread adoption of generative AI for critical processes demands careful consideration of ethical implications. Ensuring transparency in model decision-making (explainability), mitigating potential biases embedded within training datasets, and establishing robust oversight mechanisms are paramount. As these systems increasingly inform decisions with significant consequences, addressing concerns around accountability and fairness will be crucial to fostering trust and realizing the full potential of AI Deviation Management across various sectors.
The manufacturing landscape is undergoing a profound shift, driven by the remarkable capabilities of generative AI. We’ve seen how it can drastically reduce investigation times for deviations, freeing up valuable engineering resources and accelerating production cycles. This isn’t just about incremental improvements; it represents a fundamental rethinking of how we approach problem-solving in complex industrial environments. The potential to proactively identify and mitigate risks before they impact output is truly game-changing, leading to increased profitability and enhanced customer satisfaction. Successfully implementing these technologies requires careful planning and a phased approach, but the rewards are undeniable. Addressing challenges related to data quality and model governance will be key for widespread adoption and ensuring reliable outcomes. A crucial aspect of this evolution lies in effective AI Deviation Management, allowing us to monitor and refine AI models continuously, guaranteeing their performance aligns with evolving operational needs and regulatory requirements. The future of manufacturing is intelligent and adaptive; embracing generative AI is no longer a question of ‘if,’ but ‘how.’ To delve deeper into the possibilities and explore how AWS can empower your journey with generative AI and robust deviation management practices, we encourage you to check out our dedicated resources here: [Link to AWS resources on generative AI and deviation management] . Start building your smarter factory today!
Explore how these tools can revolutionize your processes and unlock new levels of efficiency. The journey towards a fully automated and optimized manufacturing operation begins with informed decisions and strategic implementation. Don’t miss out on the opportunity to be at the forefront of this technological revolution.
Continue reading on ByteTrending:
Discover more tech insights on ByteTrending ByteTrending.
Discover more from ByteTrending
Subscribe to get the latest posts sent to your email.